Quick update: Pfizer trial in <5yo shows possible NEGATIVE efficacy
There is no reason for the FDA to grant an EUA and every reason not to.
In yesterday’s post, I outlined the reasons why I’m not getting any of my three children a Covid shot, pulling data from Pfizer and Moderna’s press releases. The press releases are supposed to make results look as good as possible, but no amount of Febreze can fully mask a pile of canine excrement. You can catch the scent from the press releases, but the steaming putridity is really wafting up the nostrils now with more detailed trial data released. Can FDA officials hand us this doggie bag without their eyes watering?
Please, please, if the FDA goes ahead with the EUA as they planned, do not give this experimental shot to your kids. Six children in the trial arm for ages 2-4 (post-dose 2) met at least one criterion for severe Covid compared to 1 in the placebo. One of these cases in the vaccine group was hospitalized, Pfizer trial report shows, though he did show evidence of co-infection with parainfluenza. The trial arm is twice the size as the placebo arm, but even so that is 3:1 ratio of cases meeting at least one criterion for severe Covid for recipients of the Pfizer doses over the placebo.
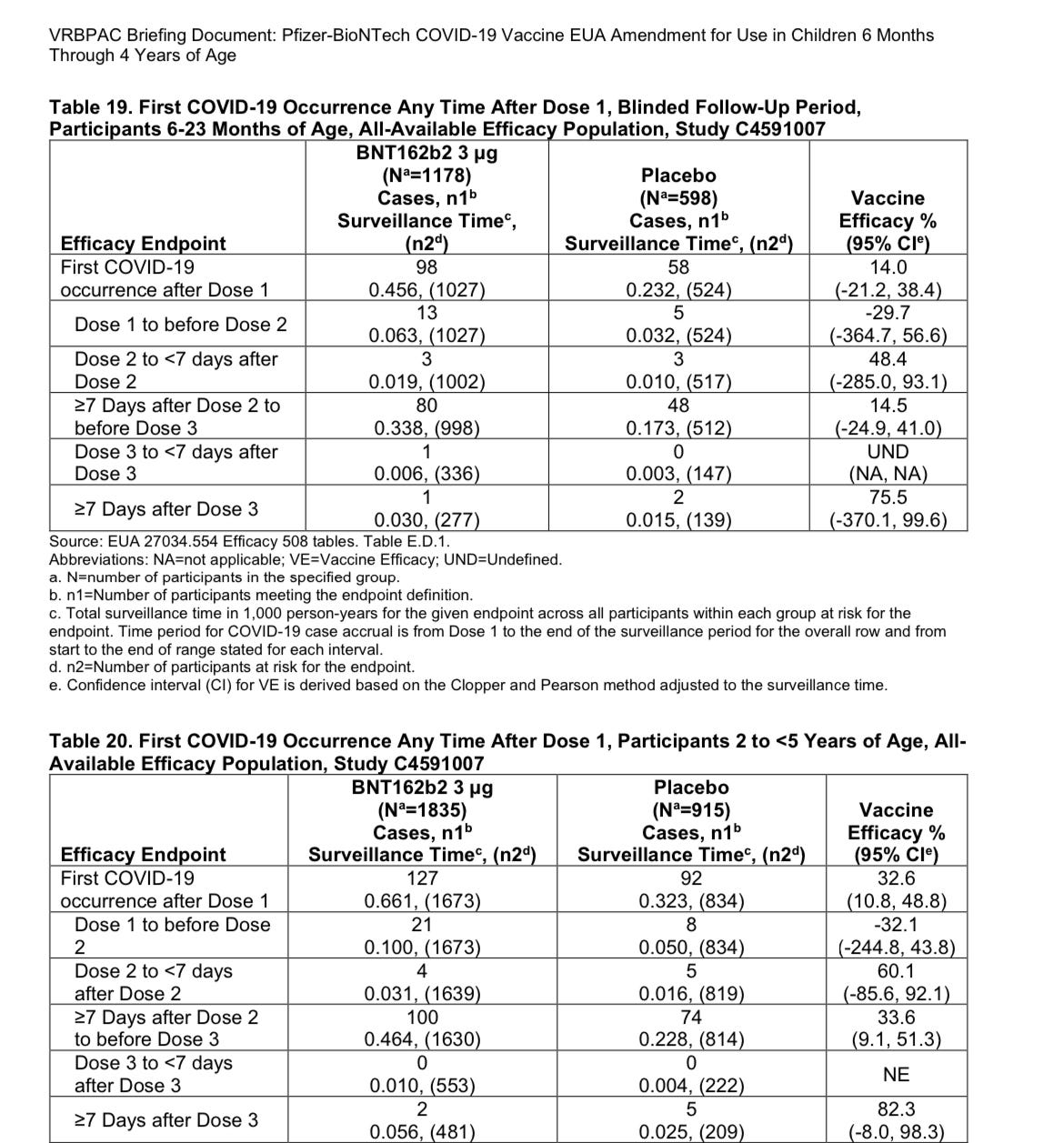
Even one week after dose 3, when protection is supposed to be highest, vaccine efficacy between 6 and 23 months old could be as low as 370% (but as high as 99.6%!). In other words, they have no idea if this product has any efficacy at all and it could be deeply negative —actually leading to more Covid infections. The lower bounds of the confidence intervals for all periods after dose 1 are negative as well. It’s worth noting the data collected by Walgreens on positive tests for SARS-COV-2 also hinted at an increased infection rate among those who received boosters (though they aren’t calling the third dose for children a booster, but part of the primary series), showing a test positivity rate of 23.7% within 5 months after their third dose (as of April 22).
While the infection rates in the Pfizer trial for those aged 2-4 don’t look quite as awful in comparison to those in the 6-23 months group, the lower bounds of the confidence interval is -8%. FDA guidance (though non-binding) issued March 31 states that an EUA would be considered only if the single point efficacy is greater than 50% and the lower bound is greater than 30%. It doesn’t take a data scientist to understand this doesn’t even come close.
Why does the FDA even bother releasing guidance if it’s going to chuck it out the window in favor of turning children into a cash cow for mega corporations with histories of fraud? And why is it that no one in the mainstream media seems to care that the FDA appears poised to brazenly break its promise to only authorize shots with at least 50% efficacy?
Moreover, guidance further states that trials should capture at least 5 cases of “severe Covid-19” in the placebo group as “generally sufficient” to determine that the vaccines are not causing enhanced respiratory disease (ERD). Instead, the cases Pfizer has indicated met at least one criterion for severe Covid in those aged 2-4 were mostly in the vaccine group, and only one was in the control. The only child hospitalized was in the vaccine group. I don’t know whether this is evidence of ERD but it certainly isn’t an assurance that there isn’t any.
Recall that once it became evident the vaccines do not “stop the spread” of SARS-COV-2, the fallback justification for mass injections was that it protects against severe Covid, and fewer cases of severe Covid means our hospitals won’t be overwhelmed. There is no evidence here that the vaccine protects young children against severe Covid, so why grant an EUA (especially considering it is less of a threat to children than seasonal influenza and produces far fewer hospitalizations). It might even be enhancing Covid symptoms. I’m not a doctor or a scientist, but any reasonable person can conclude it is not in children’s best interest to get them injected with this product, it is in Pfizer’s best interest. And if the FDA goes ahead with authorizing it, we know whose interests they really care about, and we’d do well to never, ever forget it.